What is Radiation Sterilization?
Sterilization: What is it?
Any procedure that eliminates, destroys, or deactivates all life is called sterilization. The word “sterile,” which denotes the total absence of live microorganisms or microbes with the capacity to proliferate, is associated with sterilization. As a result, after being put in their final packing, sterile products that go through sterilization are frequently thermally or chemically sterilized. Any bacteria found within the products (obtained during manufacture and packaging) are eliminated by chemical or heat sterilization. Terminal sterilization is the process of sterilizing a product using heat or chemicals after it has been packaged. Information on radiation sterilization, gamma sterilization, sterilization conveyors, and radiation sterilization businesses is included in the article below.
What does radiation sterilization mean?
A non-thermal sterilizing technique called radiation uses ultraviolet light, beta particles (an electron beam), or gamma radiation to kill germs in a product. Radiation is the only sterilization technique that doesn’t require high temperatures, other from sterile filtration. For items that cannot be sterilized using heat or chemicals, radiation is a great substitute.
What can be sterilized by radiation?
The same materials that can be sterilized by gaseous procedures are used to make radiation-compatible products. Sterilized materials and devices often include powders, heat-labile materials, and plastics. Microorganisms’ nucleoproteins are harmed by radiation. Therefore, it is not advised to use radiation on biologics.

How Does Radiation Sterilize Products?
Sterilization is simply the process of exposing a product to UV light, beta particles, or gamma radiation. Although it is not regarded as a fundamental sterilizing method, microwave radiation is occasionally utilized for empty glass containers. Because of its deep penetration, electromagnetic gamma radiation is the most efficient radiation technique. For gamma radiation, cobalt 60 high-energy photons are frequently employed. As ionizing radiations, beta particles are less penetrative and non-electromagnetic. Extremely high energy levels and the beta particles (electron beams) required for beta particle radiation are produced by mechanically accelerating strontium 90. It is also possible to utilize ultraviolet light, but only for surface sterilization. Ultraviolet light has too low an energy level to pierce materials. Although beta particle (e-beam) radiation is also utilized for product terminal sterilization, gamma sterilization is the most reliable method. Gamma sterilization is used by the majority of sterilization businesses.
The amount and duration of radiation exposure determine how effective the radiation is. Sterilization validations are conducted using a 12-D sterilization overkill technique. The 12-D represents giving the most resistant microbial spore a dosage of radiation that is enough to cause a 12-log decrease in its D value. Keep in mind that dosage, not time, is used to get the D value for radiation. Bacillus pumulis, the most radiation-resistant bacterial spore, often has D values between 1.7 and 2.0 megarad (mRad). A typical radiation exposure is 25 mRad, which is more than 12 times the D value of B. pumulis. Dosimeters are positioned at key points throughout the sterilizing procedure to track the radiation doses that products receive. As items undergoing sterilization are moved from beginning to end, the overall radiation dosage is not evenly distributed when a product passes through a radiation sterilization conveyor (see Figure 1 below). Conveyor areas closest to the gamma radiation source have the highest percentages of total radiation. A product may receive a total radiation exposure of 25 mRad. To prevent administering an excessive or harmful dose of radiation at any one point during sterilization, the radiation dosage is nevertheless dispersed throughout the conveyor system.
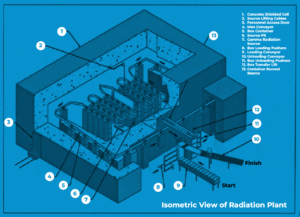
Figure 1: Radiation Sterilization Conveyor Schematic
Radiation sterilization-related factors include:
- The item being sterilized’s D value for the biological indicator or bioburden level
- Strength of radiation
- Rate of radiation dosage
- Speed of the conveyor
Radiation Sterilization: What radiation-related issues exist?
The chemical changes that occur in medication molecules and excipients when exposed to high-energy gamma radiation are not well understood. Additionally, compared to other technologies like ethylene oxide gas, there aren’t many gamma radiation or e-beam sterilization firm facilities available for bulk product sterilization. Additionally, the possibility of radiolytic byproduct generation (such as *OH) during the radiation sterilization of completed goods or active pharmaceutical ingredients (APIs) raises concerns because it may affect the raw material, API, or product packaging system.
Radiation Sterilization: In brief
All things considered, radiation is a substitute for conventional heat and chemical sterilizing techniques. A cold sterilization technique called radiation exposes microorganisms to ultraviolet, beta, or gamma rays (e-beams) to destroy them. The 12-D overkill approach is used for sterilization validations. Overall, make sure the contract testing company you select can offer the right sterilizing validations for your product requirements.







