F Value Calculations for Sterilization of Medical Devices
F Value Calculations: What is Sterilization?
Any procedure that eliminates, destroys, or deactivates all life is called sterilization. The word “sterile,” which denotes the total lack of living bacteria or viruses with the capacity to proliferate, is synonymous with sterilization. As a result, heat, chemicals, or radiation are frequently used to sterilize sterile objects. Sterilization eliminates any microbes that may have been present in the manufactured goods. After the product is put in its final packing, it is sterilized using dry heat, steam, ethylene oxide, vapor, liquid phase, and radiation. Terminal sterilization is the final step in the sterilization process after manufacture. To find out which sterilization technique is best for your medical device, please refer to our sterilization comparison chart.
What are the dynamics of microbial growth and death?
Microbial growth is exponential. Low-level contamination is not often easy to spot right away. It takes some time for the contaminating microorganisms to proliferate to the point where measurements may be made to confirm that contamination occurred. Depending on the microbial species, product storage temperature, available nutrients, and other variables that affect the kinetics of microbial growth, this lag phase can last anywhere from a few minutes to several years. Relevant lag durations for sterilization range from a few minutes to many hours. Once growth begins, it moves along rather quickly. A normal bacterial cell, for instance, may create more than 1,300,000 cells in just eight hours! When vital nutrients run out, oxygen transport is impeded, or harmful metabolites build up, microbial growth eventually reaches a stationary phase. There are roughly 10,000,000 cells per milliliter in the population where stationary phases are present.
The kinetics of microbial death are exponential, just like those of growth. A kinetic plot of microbial death is shown in Figure 17-2 below. Values and constants that characterize the kinetics of microbial death for organisms can be computed using microbial death plots. The bioburden (the starting microbial population), D value, Z value, and F value are the most often utilized quantities and constants in microbial death kinetic investigations. You can find the link between Bioburden, D value, and Z value HERE.
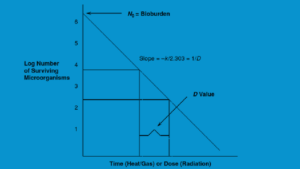
Figure 17-2: Kinetics of microbial demise
How is a F value determined, and what does it mean?
The F value is the amount of time that a product is lethally sterilized (at a specific temperature). Stated differently, the F value is the lethality of sterilization per unit of time. Bacterial spores and other biological indicators are frequently used to assess sterilization lethality. F values are also occasionally used in dry heat sterilizations. To ascertain minimum and overkill cycles for terminal sterilization validation procedures, the F value is nearly always used in steam sterilization. The F value, as opposed to D values, represents “equivalent time” rather than clock time. The temperature affects the equivalent time. For instance, the equivalent duration is always less than the real one if the measured temperature (T) is lower than the reference temperature (T0). In the F value equation below, Z is the thermal resistance value derived from D values at different temperatures. Lethality values are another name for Z values. The period between temperature readings is denoted by ∆t.
The following formula is used to determine F values:
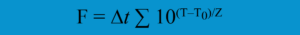
The F value compares a temperature’s microbial killing efficiency (lethality) to a reference temperature that is known to kill microorganisms. An F value of eight minutes, for instance, indicates that, regardless of the actual temperatures reached, the sterilized object was subjected to a sterilization process comparable to eight minutes at the reference temperature (for instance, 121◦C).
The information used to determine F values is shown in Table 17-3 below. Calculations for the exponential relationship between the actual temperature, reference temperature (121◦C), and a Z value of 10◦C are shown in Table 17-3 along with time and temperature data. 8.522 minutes is the result of adding together the computed exponential relationships. The F value for the data in Table 17-3 is 8.522 since the change in t (time between data collection) is one minute.

Table 17-3: Information needed to manually calculate lethality levels
The operation of F value equivalence is illustrated graphically in Figure 17-4, which displays real sterilization temperatures with time in relation to equivalent sterilization time. The computed exponential term at a specific time interval and real temperature versus time data throughout a sterilization cycle are presented in Figure 17-4. The region under the time-temperature curve, or the darkening area, is the F value result. The sterilization cycle temperature and duration are adjusted to provide a cycle with the required F value in application to sterilization validations and optimization. Sterilization cycles can be specially designed using these temperature and time adjustments, ensuring that products are sterilized to the proper F value without sacrificing their integrity.
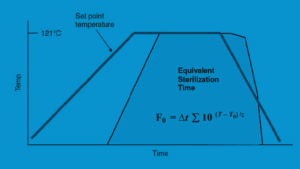
Figure 17-4: Equivalent sterilization time and sterilizer temperature-time curve comparison
The impact of common sterilizing techniques on the D value of the same microbe (B. subtilis var. niger) is shown in Table 17-2B below. As you can see, depending on the microorganism being eliminated, the efficacy of typical microbial treatments differs significantly. Microbial D values have risen throughout time, indicating the propensity of microorganisms to become resistant to the techniques used to eradicate them.

Table 17-2B: Bacillus subtilis var. niger Sterilization Treatment D Values
Another method for calculating F values is to use the biological equation (for FT) shown in Figure 17-5 below.
The starting and final microbial populations in this equation are denoted by N0 and Ns, respectively. As a function of the particular spore’s D value, the biological F value determines the F value needed to produce a spore log decrease value. Since spores are hibernating, they are more resilient to sterilizing. As an illustration of a biological F value calculation, if sterilization requires a 12-log decrease in that spore indicator organism and the D value is known to be two minutes, the minimum F value needed is 24 minutes.
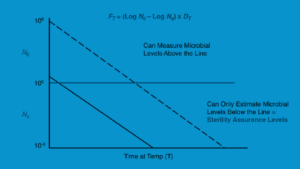
Figure 17-5: Time-dependent microbial count at a specific temperature
The microbial population plotted against time at a specific temperature (a D value graph) is depicted in Figure 17-5 above. Microbial death never reaches zero since it is logarithmic. There are places where microbial mortality is at undetectable levels because it never reaches zero. Sterilization to a sterility assurance level (SAL) is carried out because of undetectable microbiological growth levels. Ten-6, or the likelihood of one non-sterile item in a million, is an example industrial SAL value. It will take longer to attain a specific SAL if the initial microbial concentration is larger. Therefore, lowering the initial microbial concentration on goods and equipment through environmental monitoring techniques promotes quicker sterilization to the required SAL. Plots such as Figure 17-5 are used in overkill sterilization cycles to determine how long it will take to reach a specific SAL or microbial log reduction. The SAL would be 10−13 (a likelihood that one unit out of ten billion sterilized units would be contaminated) if the sample had an initial microbial contamination of 10 and the sterilization cycle resulted in a 12-log reduction of that organism.
Z values are plotted against temperature in Table 17-6 below. The lethality of a one-minute exposure at 118°C is half that of a one-minute exposure at 121°C, as this table illustrates. There is an exponential decline in the ability to destroy germs as the temperature drops from 121◦C. The fatal effect grows exponentially when the temperature rises above 121°C. Designing ideal temperatures for sterilization cycles can be aided by these temperature relationships derived from F value computations.

Table 17-6: Lethality ratios during a steam sterilization cycle as a function of temperature

An up-close view of a flask containing multiple petri dishes filled with yellow liquid. Medical device F value computations. What does a F value mean? How is an F-value determined? kinetics of microbial death. The application of F values to medical devices
In Brief
Sterilization refers to any procedure that eliminates, destroys, or deactivates all living things. In order to guarantee patient safety when using medical devices and products, sterilization is essential. The most used method of sterilizing products is steam sterilization. The lethality of sterilization per unit of time is shown by F values. The exponential relationship between the reference temperature and the actual temperature can be added up using Z values to determine F values. A biological equation that establishes the F value necessary to generate a spore log reduction value as a function of the particular spore’s D value can also be used to compute F values. F values are useful for optimizing the sterilization validation process and the sterilization cycle to achieve the required levels of sterility assurance. Make sure the contract testing company you select can offer the right sterilizing validations for your product requirements.







