Integrated Packaging & Sterilization: Ensuring Implants Arrive OR-Ready

Key Takeaways
- Orthopedic implants pose complex packaging challenges due to their weight, geometric design, and mixed materials.
- Fragmented workflows between packaging and sterilization can lead to costly delays and failures in validation.
- Integrated packaging, testing, and sterilization from the outset, resulting in improved compliance and faster time to market.
- Procedure-specific kits streamline OR setup, enhance traceability, and guarantee safe, aseptic packaging of every component.
- User-friendly and compliant packaging boosts surgeon confidence, facilitates hospital workflows, and speeds up product adoption after launch.
From Sterile to Surgical: Accelerating OR-Readiness
In today’s orthopedic market, Original Equipment Manufacturers (OEMs) are under pressure to deliver finished implants that are not only sterile but also ready for the operating room. Achieving this level of readiness requires more than just high-quality materials and manufacturing; it demands an integrated packaging and sterilization strategy from the beginning.
Unfortunately, packaging and sterilization are often treated as separate tasks managed by different vendors or addressed late in development. This fragmented approach can result in risks, delays, and costly rework. Millstone Medical Outsourcing eliminates these silos with a Tier 1 model that integrates packaging design, cleanroom assembly, in-house testing, and sterilization planning under one roof. By aligning these critical functions, Millstone ensures that sterile orthopedic devices meet regulatory standards and are ready for immediate use in the operating room.

Challenges in Packaging and Sterilizing Orthopedic Implants
Orthopedic implants present unique packaging challenges that require higher precision and protection than standard medical devices. These implants are often heavy, sharp, irregularly shaped, and composed of mixed materials that respond differently to sterilization methods.
- Heavy and Bulky Devices: Require reinforced sterile barrier systems and stronger seals to pass drop testing and resist transit stress. Standard pouches or trays may not be sufficient to maintain integrity.
- Irregular Shapes and Sharp Edges: Increase the risk of punctures or compromised sterility. Custom-molded trays and protective inserts are often needed to stabilize implants and protect barrier materials.
- Sensitivity of Mixed Materials: Many implants consist of metals, polymers, or ceramics, each with different sterilization profiles. For instance, some polymers may degrade under gamma radiation or absorb ethylene oxide (EO) gas, necessitating careful planning regarding sterilization methods and packaging compatibility.
- Regulatory Demands: Packaging systems must comply with ISO 11607 standards and support a validated sterilization process that achieves a 10⁶ Sterility Assurance Level (SAL). Under the EU Medical Device Regulation (MDR), OEMs are more responsible for delivering pre-sterilized implants that are ready for clinical use.
These challenges are amplified when packaging and sterilization are managed in isolation. Ensuring sterility and readiness starts by aligning both from the beginning.
Siloed Processes Lead to Delays and Compliance Risks
Fragmentation in packaging, testing, and sterilization introduces unnecessary risks. When OEMs manage these processes separately, often involving multiple vendors, they encounter additional challenges:
- Disconnect Between Design and Sterilization: Packaging designed without considering sterilization can fail during ethylene oxide (EO) or gamma processing due to material incompatibility or inadequate sterilant penetration.
- Late-Stage Validation Failures: Issues such as packaging breakdowns, seal failures, or non-compliance with EO residual limits can disrupt approval timelines.
- Extended Timelines: Each vendor handoff introduces communication barriers and slows the development cycle. Multiple rounds of rework can result in product launch delays of weeks or even months.
- Diluted Accountability: When vendors operate in silos, visibility and ownership of outcomes diminish, increasing the risk of non-compliance.
When packaging and sterilization processes are disconnected, OEMs face late-stage design failures, prolonged validation cycles, and heightened regulatory risks. Misaligned materials, incompatible barrier systems, or failures caused by sterilization can necessitate returning products to the design phase, costing valuable time.
With market pressures and global regulatory changes, there is a growing need for tighter coordination. OEMs that continue to rely on fragmented workflows often struggle to meet aggressive timelines and first-time validation goals. Integration is no longer optional; it is essential for success.
Integrating Packaging Design, Testing, and Sterilization from Day One
Millstone’s integrated approach aligns packaging design, cleanroom assembly, testing, and sterilization strategies. This streamlines compliance and accelerates the speed to market. Millstone collaborates with OEMs early in the product development process to ensure that all critical aspects of sterile packaging are aligned:
- Designing for Sterility: Millstone engineers collaborate early to select materials and barrier systems optimized for sterility, usability, and the chosen sterilization method. We consider factors such as ethylene oxide (EO) permeability, gamma tolerance, and the impact on shelf life right from the start.
- Designing for Assembly and Kitting: Orthopedic kits often contain screws, implants, tools, and instructions for use (IFUs). Millstone configures and assembles these components into procedural kits designed for aseptic presentation and efficiency in the operating room.
- Concurrent Testing and Iteration: Millstone’s in-house testing, including assessments of seal strength, dye penetration, bubble emission, and accelerated aging, allows for quick, data-driven adjustments during development.
- Sterilization Validation Planning: We help medical device manufacturers plan and execute EO, gamma, or vaporized hydrogen peroxide (VHP) sterilization validation strategies by ISO 11135 and ISO 11137. Aligning these strategies with packaging design from the start reduces the likelihood of revalidation and ensures compliance with sterility assurance level (SAL) requirements.
Real-time collaboration fosters faster feedback loops, reduces development friction, and increases the likelihood of successful first-time validation. Millstone’s integrated approach minimizes validation failures, shortens timelines, and enhances the probability of first-pass success, enabling OEMs to deliver sterile, market-ready implants to customers more quickly.
Kitting and Complex Assembly: Essential for Ensuring Implants Arrive OR-Ready
Hospital staff depend on quick and precise setups in the operating room. Millstone’s kitting and sterile assembly solutions enable OEMs to provide procedural surgical kits. Whether for spine, trauma, or extremities, Millstone customizes and assembles devices into formats that are ready for the OR, delivering several benefits:
- Enhanced aseptic presentation
- Simplified inventory management and tracking
- Reduced risk of missing components
- Support for Unique Device Identification (UDI) compliance and audit readiness
These services streamline hospital workflows and strengthen the OEM’s reputation for quality and clinical support.
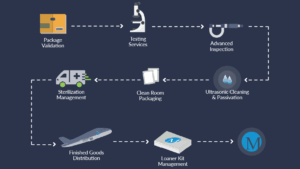
Millstone’s Tier 1 Model: Packaging and Sterilization Under One Roof Ensuring Implants Arrive OR-Ready
Millstone’s Tier 1 service model consolidates all critical services under one roof, including cleanroom packaging, labeling, assembly, in-house testing, sterilization coordination, and final distribution. This approach streamlines project execution through a single purchase order.
- ISO 7 Cleanrooms: Explicitly designed for complex orthopedic assembly and sterile barrier packaging.
- Full-Service Validation Labs: These labs conduct ISO 11607 testing, ethylene oxide (EO) residual analysis, bioburden testing, and accelerated aging assessments.
- Sterilization Expertise: Provide testing and coordination for EO, gamma, and vaporized hydrogen peroxide (VHP).
- Complex Kitting & Assembly: Multi-component systems—such as implants, screws, and trays—are assembled, labeled, and validated in-house to ensure usability and regulatory compliance.
This model reduces vendor complexity, eliminates rework, and supports flexible inventory management. OEMs gain visibility, accountability, and speed—all while meeting stringent global compliance standards.
Millstone’s Tier 1 model brings together everything OEMs need to deliver finished, sterile orthopedic products:
- Cleanroom Packaging: Operate in ISO 7 facilities and GMP-controlled environments.
- Sterile Barrier System Design: Offer validated tray and pouch configurations.
- In-House Testing: Execute ISO 11607-compliant tests for seal strength, barrier integrity, and aging.
- Sterilization Strategy Support: Advice on material compatibility, aeration cycles, and load configuration.
- Regulatory & Quality System Alignment: Maintain a unified process under one Quality Management System (QMS).
By operating under a single purchase order and centralized quality framework, Millstone eliminates handoffs, accelerates approval timelines, and ensures complete chain-of-custody visibility.
Why Ensuring Implants Arrive OR-Ready Drives Adoption and Strategic Advantage
OR-ready packaging enhances surgical efficiency and fosters adoption in a competitive orthopedic market. When implants are delivered pre-assembled, clearly labeled, and packaged for aseptic presentation, surgical teams gain immediate confidence in the product and the original equipment manufacturer (OEM) behind it.
Hospitals prioritize solutions that reduce setup time, minimize risk, and ensure traceability. An implant kit that is easy to open, intuitive to handle, and compliant with Unique Device Identification (UDI) requirements is more likely to win over surgeons, be included in preference cards, and gain institutional approval. These practical usability factors often influence long-term adoption, repeat purchases, and brand loyalty towards the OEM.
Therefore, OEMs seeking to compete in today’s orthopedic landscape must go beyond simply providing sterile products—they must be strategic. Integrated packaging and sterilization services are no longer optional; they have become a competitive advantage.
Millstone’s Tier 1 model enables OEMs to launch products more quickly, reduce rework and compliance risks, and deliver sterile, efficient, and OR-ready implants. From packaging design to final sterilization, Millstone ensures that every step is aligned, so everything functions as intended when the tray opens in the operating room.






