Streamline Medical Device Packaging to Get Ahead of Regulatory Demands
Key Takeaways
- Regulators have raised the bar – FDA and EU now require deeper proof of medical device packaging integrity, sterility, and shelf-life performance, even for legacy devices.
- Validation complexity is growing – OEMs face increased sample sizes, expanded documentation, and greater demand for end-to-end traceability.
- Non-sterile is not exempt – Medical device packaging for non-sterile devices must also protect against damage and contamination across the product lifecycle.
- Speed to market depends on packaging readiness – With Millstone’s Tier 1 model, OEMs gain a strategic partner to meet today’s standards and tomorrow’s timelines.
- Millstone simplifies compliance – Our integrated services reduce handoffs and accelerate validation with in-house testing, cleanroom assembly, and pre-validated formats.
Medical Device Packaging in a New Era of Compliance Oversight
The post-COVID era has reshaped more than just supply chains. It has redrawn the regulatory roadmap for medical device packaging. From updated ISO standards to evolving FDA expectations, Medical device original equipment manufacturers (OEMs) are facing unprecedented scrutiny around sterile barrier systems, biocompatibility, and shelf-life validation.
What used to be a linear, box-checking exercise in packaging validation is now a dynamic, high-stakes process requiring deep documentation, system-level traceability, and greater alignment with risk-based quality management. And it’s not just sterile packaging under the microscope. Non-sterile systems must also prove they protect devices against damage and contamination.
For many OEMs, regulatory compliance has become a constant source of pressure, draining time, budgets, and internal bandwidth. It’s no longer about checking a box or passing a single validation protocol. Today’s landscape demands that OEMs navigate a shifting web of international and regional regulations, each imposing new rules on packaging integrity, material safety, environmental impact, and other factors.
These expectations don’t stop at sterility. They stretch into biocompatibility, labeling precision, distribution testing, and shelf-life reliability. Meanwhile, non-packaging compliance areas, such as cybersecurity mandates, data governance, and ESG reporting, are compounding the complexity. The result is slower launches, rising costs, and growing regulatory risk.
How Medical Device Packaging Standards Have Changed Since 2020
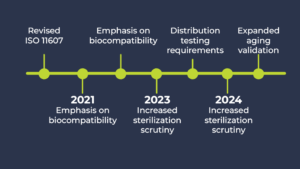
Medical device packaging standards haven’t just evolved—they’ve hardened. Since 2020, the FDA and the EU MDR have intensified their focus on packaging and labeling as critical components of device safety, thereby elevating expectations for validation, documentation, and performance.
Key changes include:
- Updated ISO 11607 (Packaging for Terminally Sterilized Medical Devices):
The global benchmark for sterile packaging validation was revised in 2019 and 2020, introducing stricter requirements for demonstrating seal integrity, aging, and real-world performance.
- New Emphasis on Biocompatibility:
Packaging materials that come into contact with the device or could leave residues are now subject to biological evaluation under ISO 10993-1 (Biological Evaluation of Medical Devices – Part 1: Evaluation and Testing within a Risk Management Process).
- Distribution and Transit Testing:
ASTM standards, such as ASTM D4169 (Performance Testing of Shipping Containers and Systems), are now essential for demonstrating that packages can withstand the shocks, vibrations, and temperature fluctuations of real-world logistics.
- Sterile Barrier Reliability: Regulators expect 95% confidence at 95% reliability that the package remains sterile, which significantly increases the required sample sizes and scrutiny.
- Non-Sterile Packaging Oversight: Packaging must protect devices, even those not terminally sterilized, from physical damage and contamination throughout the product’s lifecycle.
What does this mean for OEMs? OEMs face longer validation timelines, higher documentation demands, and the need to re-evaluate previously approved packaging formats.
What Regulators Are Really Looking For
Today’s regulatory focus goes beyond simple compliance. Agencies are looking for:
- Integrated Risk Management: ISO 14971 (Application of Risk Management to Medical Devices) requires validation plans to align with its guidelines, ensuring every packaging decision prioritizes patient safety.
- End-to-End Testing:
- Seal strength (ASTM F88/F88M)
- Bubble emission and dye penetration (ASTM F2096, ASTM F1929)
- Accelerated aging and shelf-life studies (ASTM F1980)
- Shipping simulation and drop tests (ASTM D4169, ASTM D5276)
- Residual sterilant analysis for EO (per ISO 10993-7)
- Seal strength (ASTM F88/F88M)
- Biocompatibility Justification: Packaging materials, including inks, adhesives, or tray plastics, must be demonstrated to be non-reactive to ensure they do not transfer to the device.
- UDI and Labeling Compliance: Proper serialization and device labeling are now regulated by ISO 15223-1 (Symbols for Medical Device Labels) and ISO 20417 (Information to be Supplied by the Manufacturer). Label placement and content are part of packaging validation.
If you can’t prove it, it didn’t happen. That’s the new reality of medical device packaging.
Why It’s Getting Harder to Keep Up
For device manufacturers, these changes come with growing pressure and complexity:
- Legacy Device Revalidation: Devices that have been on the market for years may now fall short of new expectations, triggering the need for retrospective validation.
- Increased Documentation Load: Regulators expect an ecosystem of proof, including risk-based rationales, sample size justifications, environmental conditioning data, and end-of-life testing.
- Interpretation Challenges: Even the most experienced RA/QA teams can struggle to translate evolving standards into concrete action plans. What counts as sufficient evidence? How do packaging materials interact with device sterility?
- Resource Bottlenecks: Most in-house teams don’t have the cleanroom space, testing equipment, or validation expertise to generate all the required data. Outsourcing becomes essential.
Many OEMs encounter a critical moment: their device is ready, but the packaging isn’t. What looks complete on paper often reveals problems during testing—seals fail, materials degrade, or sterile barriers can’t withstand distribution stress. Instead of a swift launch, teams face costly redesigns and repeated validation cycles. These challenges often stem from packaging being developed too late, in isolation, or without essential technical input. With internal resources stretched thin and few teams having the necessary cleanroom access or regulatory expertise, quick fixes are rare.
How Millstone Helps You Stay Ahead with Compliant Medical Device Packaging

Millstone Medical Outsourcing built its integrated Tier 1 model for this moment. It combines packaging, cleanroom assembly, and in-house ISO 17025-accredited testing, creating a single-source validation ecosystem OEMs can rely on.
Our core solutions include:
- Comprehensive Packaging Validation: Millstone executes complete ISO 11607-compliant test protocols, including:
- Seal integrity testing
- Transit simulation per ASTM D4169
- Aging studies and shelf-life data generation
- Sterile barrier performance
- Seal integrity testing
- Biocompatibility Testing: Through ISO 10993-1 evaluations, we ensure that packaging materials meet safety standards and do not compromise device performance or patient safety.
- Cleanroom Packaging & Assembly: With an ISO 7 cleanroom space and validated procedures, we minimize contamination risk and enhance packaging precision at every step.
- Pre-Validated Packaging Formats: Our tray and pouch systems come with validated sealing, aging, and integrity data. OEMs can often leverage these formats to reduce regulatory timelines and testing costs.
- Expert Regulatory Support: We help interpret complex standards and develop rationales that meet FDA and EU MDR expectations, reducing the risk of rework, remediation, or launch delays.
- End-to-End Traceability: From serialization to label compliance, we help OEMs meet traceability requirements across global markets.
The result? Faster compliance. Lower risk. Fewer handoffs. Greater confidence.
The Millstone Difference: Speed Without Compromise
Unlike fragmented outsourcing models, Millstone unifies the entire packaging lifecycle under one roof:
- Single Partner, Single PO: Cleanroom packaging, labeling, testing, sterilization support, and final release are all managed by one trusted team.
- Built-In Compliance: Millstone ensures that every procedure, protocol, and output aligns with ISO and FDA regulations. No more guessing.
- Scalable Across Products and Markets: Whether you’re launching a new Class III device or updating packaging for a legacy product across multiple SKUs, Millstone has the infrastructure and regulatory clarity to scale with you.
- Strategic Speed to Market: Our clients don’t just validate—they validate early, thoroughly, and efficiently. That’s how we help you launch with fewer delays and stronger audit resilience.
By integrating packaging, testing, and sterilization into a single workflow, Millstone eliminates the friction that slows down launches and increases risk. Millstone doesn’t just check boxes; we help you build a packaging system that performs under pressure, withstands regulatory scrutiny, and supports your team from development through distribution. When every step works together, speed does not come at the cost of quality. It becomes a strategic advantage.
Medical Device Packaging That Turns Regulation into Readiness
Today, medical device packaging goes beyond mere protection. It demands precision, validation, and readiness, especially within an increasingly stringent and time-sensitive global regulatory landscape. Effective packaging can often be the critical factor between timely approval and costly delays.
Millstone Medical Outsourcing is your partner in navigating these challenges. We offer comprehensive, end-to-end solutions, supported by our in-house laboratories and a team proficient in regulatory language. We transform intricate demands into assured implementation.
Allow Millstone to streamline your packaging approach, expedite your regulatory compliance, and ensure that every device arrives at its destination precisely as required: secure, sterile, and fully functional.







