Biocompatibility Testing for Orthopedic Combination Products
Key Takeaways
- Start testing early. Begin biocompatibility testing in the design phase to avoid late-stage failures.
- Plan for complexity. Combination products require integrated validation of device, drug, and packaging.
- Don’t rely solely on ISO 10993. Include extractables and leachables, cytotoxicity screening, and sterilization testing.
- Test sterilization impact. EO, gamma, and steam can degrade drugs or create toxicity if not validated correctly.
- Validate dual stability. Test devices to ASTM F1980 and drugs to ICH Q1A—on the same samples when possible.
- Treat packaging as part of the system. It must maintain barrier integrity, preserve the drug, and avoid interactions.
- Avoid siloed testing. Fragmented labs miss cross-interactions and cause regulatory delays.
- Choose integrated testing. Millstone brings all testing under one roof to accelerate timelines and reduce risk.
Biocompatibility Isn’t a Checkbox Anymore
Orthopedic devices are rapidly advancing beyond traditional metal and polymer constructs. Today, many incorporate drugs or biologics to enhance patient outcomes by combating infections, accelerating healing, or mitigating inflammation. However, these combination products introduce significant complexity.
The inclusion of a drug or biologic fundamentally changes the regulatory landscape. Devices must now satisfy not only mechanical and sterility benchmarks but also stringent pharmaceutical-level safety requirements, making biocompatibility testing considerably more challenging.
Too frequently, medical device Original Equipment Manufacturers (OEMs) approach biocompatibility as a mere checklist item. This oversight can lead to severe repercussions if not addressed strategically early in the development process. Issues such as cytotoxic reactions, extractable failures, or leachables from packaging materials can emerge late in the process, necessitating costly rework just as sterilization and packaging validation are nearing completion.
Failing to integrate biocompatibility into the product strategy from the outset directly leads to delays, retesting, and lost market time.
Understanding the Complexity of Orthopedic Combination Products
Orthopedic combination products offer innovative solutions by integrating medical devices with active drug or biologic components. These products aim to enhance clinical outcomes by providing localized treatment at the surgical site, which can reduce systemic side effects, improve healing, and strengthen the effectiveness of post-surgical care. However, their dual nature means these advanced devices face increased technical and regulatory scrutiny.
Common examples include:
- Antibiotic-coated trauma plates to prevent postoperative infection.
- Drug-eluting bone grafts that release growth factors over time.
- Bone cements infused with anti-inflammatories to manage pain.
- Resorbable polymers embedded with biologics to accelerate healing.
The introduction of a drug or biologic shifts the regulations. Regulatory bodies, such as the FDA and EU MDR, categorize these as combination devices. Manufacturers must then prove the safety of the device, the safety of the drug or biologic, and that the two components operate safely in conjunction.
That means proving:
- Biocompatibility of all materials
- Safety and efficacy of the drug or biologic
- No adverse interactions between components
- Compatibility with sterilization and packaging methods
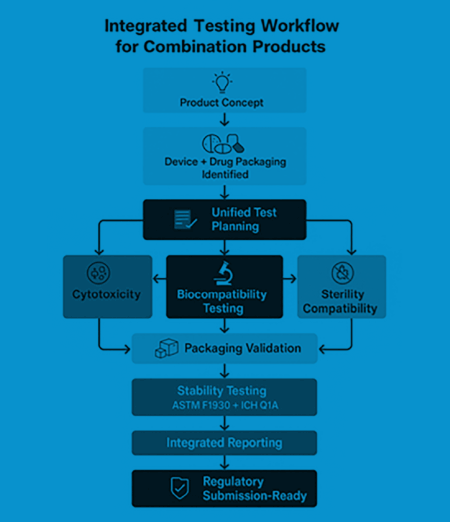
OEMs need to rethink their biocompatibility testing approach for combination products. Instead of a final checklist item, biocompatibility testing should be an early and integrated part of the development process. A unified strategy for validating the device, drug, and packaging, executed from the outset, minimizes risk, avoids costly rework, and accelerates the regulatory approval process.
New Layers of Complexity with Biocompatibility Testing
For standard implants, traditional biocompatibility testing, as outlined in ISO 10993, may be sufficient. However, it is no longer adequate for today’s combination products, which introduce a new level of complexity. These devices require validation of chemical interactions, drug stability, and packaging compatibility to ensure their safety and efficacy. OEMs who address biocompatibility late in the development process often find that their existing protocols are insufficient, leading to costly failures and regulatory issues.
Combination products require:
- Evaluation of chemical interactions between the device, drug, and packaging
- Advanced extractables and leachables (E&L) testing to uncover potential toxicity
- Cytotoxicity screening of packaging materials that may affect drug stability
- Coordination with sterility and stability protocols to ensure complete lifecycle safety
Failing to integrate biocompatibility testing early across device, drug, and packaging systems rapidly exacerbates issues for OEMs. A simple missed requirement can escalate into failed assays, costly formulation adjustments, and significant delays in submission readiness. These last-minute complications force internal teams into a reactive stance, scrambling to update sterilization protocols or revalidate packaging under pressure. Reacting late not only erodes stakeholder confidence but also depletes resources and postpones critical launch timelines.
Millstone mitigates these risks with early-stage coordination and expert oversight. With Millstone, OEMs gain access to in-house and partnered biocompatibility labs, early screening of packaging materials to rule out toxic responses, and unified protocols for extractables and leachables, sterility, and potency testing. By identifying risks proactively during the design phase, Millstone helps OEMs avoid costly rework and late-stage test failures, ensuring combination products stay on track from concept to submission.
Sterilization Complications That Can Derail Biocompatibility Testing
Sterilization is non-negotiable in medical device manufacturing—but for orthopedic combination products, it introduces a unique set of biocompatibility risks. Standard methods, such as ethylene oxide (EO), gamma irradiation, or steam, can destabilize drug compounds, alter release profiles, or chemically interact with coatings and absorbent polymers. These effects often go undetected until late-stage validation, when making changes is both costly and time-consuming.
To gain regulatory approval, OEMs must now demonstrate not just that the product is sterile, but that the drug or biologic remains stable, safe, and effective after sterilization. This dual burden significantly increases complexity.
Key concerns include:
- EO residuals absorbed by drug-eluting polymers, potentially leading to cytotoxicity or regulatory rejection
- Thermal degradation of sensitive biologics during gamma or steam sterilization can compromise their therapeutic efficacy.
- Loss of potency or altered drug-release profiles due to sterilization-induced changes in formulation or packaging material behavior
Solving these challenges takes more than trial and error or last-minute adjustments. It requires a proactive, integrated strategy—one that anticipates how sterilization will impact every element of the combination product, from the formulation to the final packaging.

Millstone helps OEMs manage all sterilization variables under one roof by:
- Simulating EO, gamma, and steam sterilization cycles tailored to each product’s materials and drug profile
- Performing post-sterilization analysis to assess residual EO, drug potency, and structural integrity
- Validating packaging and drug-device performance in real-world conditions to ensure both safety and efficacy
By addressing sterilization risks early in the development process, OEMs can avoid late-stage cycle revisions, reduce regulatory risk, and stay on track for a successful launch.
Meeting Dual Stability Standards for Combination Devices
Combination products must meet both device and drug stability standards—two separate regulatory expectations that now converge into a single, high-stakes requirement.
To satisfy regulators, OEMs must adhere to:
- ASTM F1980 for accelerated aging of medical devices and packaging
- ICH Q1A for drug stability, potency, and shelf-life evaluation under pharmaceutical conditions
Stability testing must evaluate all components of the product system, including:
- Mechanical integrity and sterile barrier of the device and packaging over time
- Potency, purity, and release rate of the drug across multiple storage conditions
- Chemical interactions between packaging materials and active drug components
Failure to test these variables together—not just in isolation—can result in failed release profiles, degraded drug function, or packaging that no longer protects or complies with regulations.
Millstone helps OEMs reduce these risks through a consolidated testing strategy that includes:
- Executing ICH Q1A and ASTM F1980 protocols in parallel to streamline timelines
- Using real-time and accelerated aging chambers with controlled temperature, humidity, and light exposure
- Testing drug potency and device functionality on the same-aged samples to uncover interdependent risks early
This integrated approach minimizes cost, compresses validation timelines, and ensures OEMs submit comprehensive, audit-ready data to both FDA and EU authorities.
Strengthen Biocompatibility Testing by Integrating Packaging Validation
In combination devices, packaging does more than protect—it interacts. Regulatory bodies treat packaging as part of the drug delivery system, requiring proof that it preserves the safety, efficacy, and shelf life of the product throughout its lifecycle.
Testing must confirm that the packaging:
- Prevents extractables and leachables from contaminating the drug
- Maintains sterile barrier integrity over time
- Survives shipping, vibration, and drop forces without compromising protection
These risks grow when packaging materials interact with active ingredients. Chemical reactions can cause cytotoxicity, degrade drug potency, or alter release profiles. That’s why isolated packaging tests are no longer enough.

Millstone helps OEMs close this gap by integrating packaging validation into the broader testing strategy. The team:
- Conducts extractables and leachables analysis during material selection
- Uses real-time and accelerated aging chambers to monitor barrier stability
- Performs mechanical simulations to ensure packaging survives handling and transit
By coordinating these tests with biocompatibility and drug stability analysis, Millstone ensures that packaging supports the entire system, not just the shelf life. OEMs gain submission-ready data and reduce the risk of late-stage surprises.
The Risk of Fragmented Testing
Many OEMs take a siloed approach to testing combination products. They send the device to one lab, the drug component to another, and outsource packaging validation separately. While this may seem efficient upfront, it often creates fragmented data sets, missed cross-interactions, and inconsistent documentation.
This disjointed strategy invites regulatory scrutiny. Without a unified testing plan, teams struggle to demonstrate how each component performs as part of a whole. Regulators flag gaps, question data integrity, and request additional testing—delaying approval and driving up costs.
Millstone eliminates these risks by bringing everything under one roof. Their integrated model provides:
- A single team coordinating device, drug, and packaging testing
- Unified test protocols that account for interactions between components
- Centralized documentation that aligns with FDA and EU MDR expectations
This cohesive approach reduces rework, accelerates regulatory submissions, and improves audit outcomes. Instead of reacting to late-stage surprises, OEMs move forward with confidence, knowing their data reflects the whole product lifecycle.
Biocompatibility Confidence Starts with Integrated Testing
Orthopedic combination products demand more than a checklist approach. The device, the drug, and the packaging must be validated not only individually but also together as an integrated system. Fragmented testing models can’t keep up with the complexity or the heightened regulatory scrutiny these products now face.
Millstone brings it all together under one roof:
- Biocompatibility testing – Cytotoxicity, sensitization, systemic toxicity
- Extractables and leachables (E&L) – Profiling drug-package interactions
- Sterility and EO residual testing – For sensitive polymers and coatings
- Stability testing – ICH Q1A and ASTM F1980 for drug and device
- Packaging validation – ISO 11607 with real-world drop and vibration simulation
- Regulatory coordination – FDA- and EU-aligned documentation from a single team
By integrating testing, OEMs not only meet but also exceed standards. That level of rigor is what it takes to launch today’s most advanced orthopedic combination products confidently.